An obsolete term for an iron-rich chlorite.
An obsolete term for an iron-rich chlorite.
An obsolete term for a poorly defined material perhaps related to vermiculite.
An obsolete term for lepidolite.
A general term for major dietary nutrients required in relatively large quantities per day. For example for human consumption, macronutrients include Ca K, Na, Mg that may be potentially provided by ingestion of clays, whereas clays in soils provide these nutrients for plants.
Cf., micronutrient
In clay science, a macropore (Figure 3) is a cavity among clay particles or aggregates with a diameter of >50 nm, in accord with IUPAC convention (Rouquerol et al., 1994). In soil science, a macropore is defined as a cavity among aggregates with a diameter of >75 μm (Soil Science Society of America, 1997). The pore volume (or pore size) distribution of clays is commonly determined by gas adsorption methods (typically H2O or N2), whereas in soil science/physics, it is usually determined by mercury intrusion porosimetry (MIP) technique.
Cf., mesopore, micropore
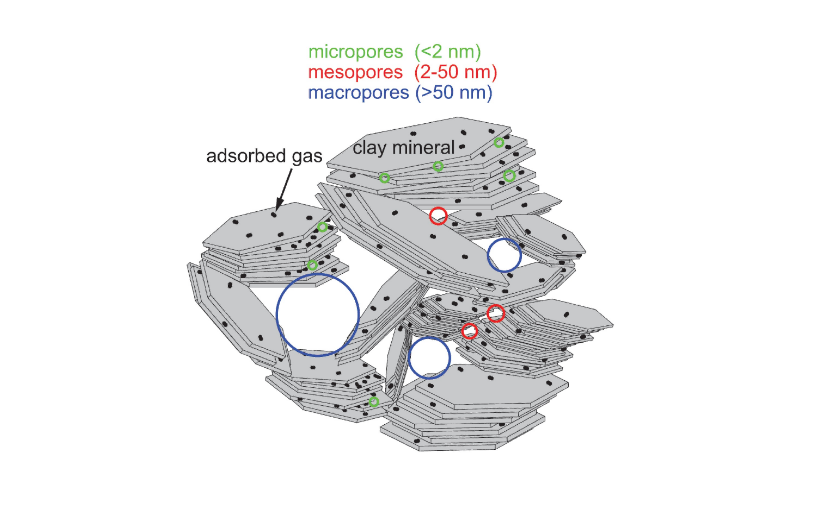
Figure 3. Schematic illustrating micropores, mesopores, and macropores in an aggregate of clay mineral particles. Whereas macropores occur between stacks of clay particles, micropores exist at the edges of clay particles and stacks, often relating to partially bent edges, translational disorder, and/or rotational disorder. Adsorbed gas molecules are illustrated by nitrogen molecules, a common gas used for gas adsorption methods.
A geological term describing magmas, igneous rocks and silicate minerals that have a relatively high concentration of magnesium and iron and a low silica concentration. Mafic magmas typically have low viscosity, when compared to felsic magmas. Mafic rocks are typically darkly colored when compared to felsic rocks.
Cf., felsic
A platy, hydrous alkali silicate of composition approximating Na2O . 14SiO2 . 9H2O with exchange properties, and which forms by precipitation from alkaline lakes rich in carbonate/bicarbonate brines, such as that found at Lake Magadi, Kenya. Suggested formulae include NaSi7O13(OH)3 . 3H2O and NaSi6O12(OH). The latter formula, which differs somewhat from the chemical ratio (due to supposed impurities), is derived from a proposed structure model (Garcés et al., 1988) based on the zeolites in the mordenite group: the model consists of continuous sheets of six-fold SiO4 rings of tetrahedra and adjacent five-fold rings pointing away from the sheet surfaces. Other structure models are possible. Kenyaite, with an approximate formula of NaSi11O20.5(OH)4 . H2O, forms under a similar environment as magadiite and is probably a layer structure also.
An obsolete term for phlogopite.
An obsolete varietal term for clintonite.
An obsolete varietal term for magnesian illite.