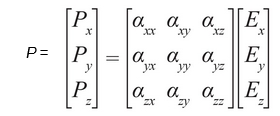The ability of an atom, ion, or molecule to become polarized (where there is a distortion of its charge distribution). For example, an anion has greater polarizability than a cation because of the tendency of the electron cloud about an anion to be easily distorted by a local electric field. Generally, an anion is unable to hold its outer electrons tightly and therefore it is more readily polarizable than a cation. For a monoatomic atom or ion and some molecules, the polarization vector P has the same direction as the applied electric field vector E, and P = αE, where α is the polarizability. If a molecule has an anisotropic polarizability, that is the polarization P may not follow the direction of the applied electric field E, the polarizability α is a symmetric tensor, and the polarization P is given as:
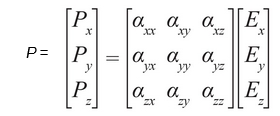
and because the polarizability tensor is symmetric: alpha(xy) = alpha(yx), alpha(xz) = alpha(zx), and alpha(yz)=alpha(zy).
Cf., polarization, dipole moment